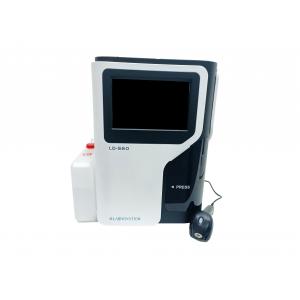
Labnovation LD-560 HbA1c Analyzer HPLC System For HbA1c Testing Dual Mode With NGSP&IFCC Certificated
Add to Cart
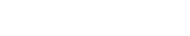
Labnovation LD-560 HbA1c Analyzer HPLC System For HbA1c Testing Dual Mode With NGSP&IFCC Certificated
Intended Use
The Fully Automated HbA1c Analyzer is used in conjunction with suitable reagents for the quantitative detection of glycosylated hemoglobin (HbA1c) in human blood.
Specification
| Model | LD-560 |
| Method | HPLC |
| Parameters | HbA1c, HbF, HbA1a, HbA1b, HbA0; HbA2 (optional) |
| Test Speed | 90 seconds per test |
| Reagents kit | Eluent A, Eluent B, Hemolysis |
| Autoloader Capacity | 20 tubes |
| Precision | CV<2% |
| Dimension | 378(L) ×380(W)×510(H)mm |
| Weight | 28kg |
Full automatic smart system
- One click start testing
- Automatic sample type recognition
- Automatic testing mode switch
Automatic scanning of barcode
Autoloader for 20 tubes
Easy to change reagents
Operation Preparation
Before operating this analyzer, please read the operation manual carefully to avoid malfunction or damage.
If the analyzer is used in a manner not specified by the manufacturer, the protection provided by the analyzer may be destroyed.
Before turning on the analyzer, the operator must check the following requirements to ensure that the system is ready.
(1) Check the remaining amount of reagents
Check whether the remaining amount of eluent A, eluent B, eluent C and hemolysis agent is sufficient.
(2) Check the waste liquid barrel
Check whether the waste liquid barrel has been emptied before starting the analyzer every day.
(3) Check the liquid tubes and power supply
Check whether the tubes of reagent and waste liquid are bent and whether the connections are reliable.
Check the analyzer for fluid leakage.
Check whether the power plug of the analyzer is securely inserted into the power socket.
(4) Check the printer
Check whether the paper roll is sufficient and whether it is installed in correct place.
Certificate
Attention
- Please make sure that this analyzer is used under the conditions specified in the instruction manual. The incorrect instruction may result in abnormal operation of the analyzer, unreliable measuring results, and possibly cause damage to parts of the analyzer or even personal injury.
- The instrument should be inspected strictly before unpacking. In order to avoid breakage in transportation, the instrument is carefully packaged. When receiving the analyzer, please carefully check if there is physical damage in the packaging. If the packaging is damaged, please contact our services personnel.
- Please always keep it upward when unloading and delivering the instrument. Never tilt or roll the instrument. The foam inside the box can prevent shock to a certain extent during the handling of the instrument. Minimize the vibration during handling. After handling, perform checks and debugging.