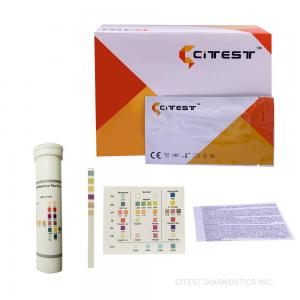
Semi Quantitative Biochemistry Test Kit Convenient Urine Adulteration Test Dipstick
Brand Name:Citest
Certification:CE
Model Number:DAT-111
Minimum Order Quantity:N/A
Delivery Time:2-4 Weeks
Place of Origin:-
Contact Now
Add to Cart
Verified Supplier
Location:
Hangzhou Zhejiang
Address:
170-422, Richards Street, Vancouver BC V6B 2Z4, CANADA
Supplier`s last login times:
within 28 hours
Shipping
lt's easy to get a shipping quote! Just click the button below and complete the short form.
Get Shipping Quote
Product Details
Company Profile
Product Details
Adulteration Dipstick, Biochemistry Reagents , Convenient Urine Test Dipstick
The Urine Adulteration Test Dipsticks are a semi-quantitative color comparison screen for the detection of Creatinine, Nitrite, Glutaraldehyde, pH, Specific Gravity, Oxidants and Pyridinium Chlorochromate in human urine.
Each of the plastic Dipsticks contains six chemically treated
reagent pads. One minute following the activation of the reagent
pads by the urine specimen, the colors that appear on the pads can
be compared with the printed color chart on the canister. The color
comparison provides a semi-quantitative screen for Creatinine,
Nitrite, Glutaraldehyde, pH, Specific Gravity, Oxidants and
Pyridinium Chlorochromate in human urine, which can help assess the
integrity of the urinespecimen.
How to use?
Allow the Dipstick, urine specimen, and/or controls to reach room
temperature (15-30ºC) prior to testing.
1. Remove the Dipstick from the closed canister and use it within
one hour. Immediately close the canister tightly after removing the
required number of Dipstick(s). Completely immerse the reagent
areas of the Dipstick in fresh, wellmixed urine and immediately
remove the Dipstick out of the urine specimen to avoid dissolving
the reagents. See illustration 1 below.
2. Hold the Dipstick in a horizontal position and immediately bring
the edge of the Dipstick into contact with an absorbent material
(e.g. a paper towel) to avoid mixing chemicals from adjacent
reagent areas and/or soiling hands with urine. See illustration 2
below.
3. Read results in 1 minute by comparing the reagent areas to the
corresponding color blocks on the Color Card. Hold the Dipstick
close to the color blocks and match carefully. Do not interpret
results after 4 minutes.
Order Information:
| Cat. No. | Product Description | Specimen | Format | Kit Size | Cut-Off | Status |
| DAT-111 | Adulteration Dipstick | Urine | Dipstick | 25 T | / | CE |

Semi Quantitative Biochemistry Test Kit Convenient Urine Adulteration Test Dipstick
Inquiry Cart
0