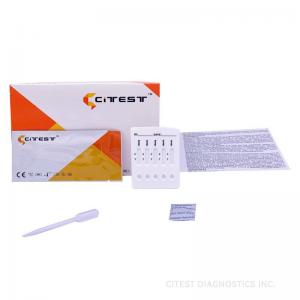
15 Minutes HBV Combo Rapid Test Cassette HBsAg Rapid Test Dipstick
Brand Name:Citest
Certification:qualified
Model Number:IHB-355/IHBSG-301/IHBSG-302
Minimum Order Quantity:N/A
Delivery Time:2-4 Weeks
Place of Origin:-
Contact Now
Add to Cart
Verified Supplier
Location:
Hangzhou Zhejiang
Address:
170-422, Richards Street, Vancouver BC V6B 2Z4, CANADA
Supplier`s last login times:
within 28 hours
Shipping
lt's easy to get a shipping quote! Just click the button below and complete the short form.
Get Shipping Quote
Product Details
Company Profile
Product Details
HBV Combo Rapid Test Cassette, HBsAg Rapid Test Dipstick/Cassette
A rapid test for the qualitative detection of Hepatitis B Surface
Antigen (HBsAg), Hepatitis B Surface Antibody (HBsAb), Hepatitis B
Envelope Antigen (HBeAg), Hepatitis B Envelope Antibody (HBeAb),
and Hepatitis Core Antibody (HBcAb) in serum or plasma.
For professional in vitro diagnostic use only.
| Product features | Parameters |
| Principle | Chromatographic Immunoassay |
| Format | Dipstick, Cassette |
| Specimen | WB / S / P |
| Reading Time | 15 minutes |
| Pack | 25 T/ 40 T/ 50 T |
| Storage Temperature | 2-30°C |
| Shelf Life | 2 Years |
INTENDED USE
The HBV Combo Rapid Test Cassette is a rapid chromatographic
immunoassay for the qualitative detection of HBsAg, HBsAb, HBeAg,
HBeAb and HBcAb in serum or plasma.
SUMMARY
Chronic hepatitis B is a serious, debilitating illness that can
cause cirrhosis of the liver, liver cancer and death. Chronic
hepatitis B is the main cause of liver cancer and the tenth leading
cause of death worldwide, with 400,000,000 people infected with the
virus. Every year, one million people worldwide are expected to die
from this infection.
Most people fight off the infection themselves, but approximately
5-10 percent of those infected with the virus become carriers, and
an additional 5-10 percent of those infected each year will
progress to chronic liver disease, cirrhosis and possibly liver
cancer.
The HBVCombo Rapid Test Cassette (Serum/Plasma) is a rapid test to
qualitatively detect the presence of HBsAg, HBsAb, HBeAg, HBeAb and
HBcAb in serum or plasma without the use of an instrument.
PRINCIPLE
HBsAg and HBeAg
The HBsAg and HBeAg tests are qualitative, two-site sandwich
immunoassays for the detection of HBsAg or HBeAg in serum or
plasma. The membrane is pre-coated with anti-HBsAg or anti-HBeAg
antibodies on the test line region of the strip. During testing,
the serum or plasma specimen reacts with the particle coated with
anti-HBsAg or anti-HBeAg antibodies.2The mixture migrates upward on
the membrane chromatographically by capillary action to react with
anti-HBsAg or anti-HBeAg antibodies on the membrane and generate a
colored line. The presence of this colored line in the test line
region indicates a positive result,
while its absence indicates a negative result.
HBsAb
Hepatitis B surface Antibody (HBsAb) is also known as
anti-Hepatitis B surface Antigen (anti-HBs). This test is a
qualitative, lateral flow immunoassay for the detection of HBsAb in
serum or plasma. The membrane is pre-coated with HBsAg on the test
line region of the strip.
During testing, the serum or plasma specimen reacts with the
particle coated with HBsAg. The mixture migrates upward on the
membrane chromatographically by capillary action to react with
HBsAg on the membrane and generate a colored line. The presence of
this colored line in the test line region indicates a positive
result, while its absence indicates a negative result.
HBeAb and HBcAb
Hepatitis B envelope Antibody (HBeAb) is also known as
anti-Hepatitis B envelope Antigen (anti-HBe). Hepatitis B core
Antibody (HBcAb) is also known as anti-Hepatitis B core Antigen
(anti-HBc). These tests are immunoassays based on the principle of
competitive binding.
During testing, the mixture migrates upward on the membrane
chromatographically by capillary action. The membrane is pre-coated
with HBeAg or HBcAg on the test line region of the strip. During
testing, anti-HBe antibody or anti-HBc antibody, if present in the
specimen, will compete with particle coated anti-HBe antibody or
anti-HBc antibody for limited amount of HBeAg or HBcAg on the
membrane, and no line will form in the test line region, indicating
a positive
result.
A visible colored line will form in the test line region if there
is no anti-HBe antibody or anti-HBc antibody in the specimen
because all the antibody coated particles will be captured by the
antigen coated in the test line region.
To serve as a procedural control, a colored line will always appear
in the control line region indicating that proper volume of
specimen has been added and membrane wicking has occurred.
REAGENTS
The test cassette contains anti-HBsAg particles, HBsAg particles,
anti-HBeAg particles, HBcAg particles respectively and anti-HBsAg,
HBsAg, anti-HBeAg, Anti-HBcAg coated on the membrane respectively.
DIRECTIONS FOR USE
Remove the test cassette from the sealed foil pouch and use it as
soon as possible. Best results will be obtained if the assay is
performed within one hour.
Place the test cassette on clean and level surface. Hold the
dropper vertically and transfer 3 full drops of serum or plasma
(approx.75ul) to each sample well of the test cassette
respectively, then start the timer. Avoid trapping air bubbles in
the specimen well. See the illustration below.
Wait for the red line to appear. The result should be read at 15
minutes. Do not interpret the results after 20 minutes.
INTERPRETATION OF RESULTS
(Please refer to the illustration above)
Warning: Do not interpret all 5 tests with the same criterion.
Carefully follow the directions below.
HBsAg, HBsAb, HBeAg
POSITIVE:* Two distinct colored lines appear. One colored line
should be in the control region (C) and another colored line should
be in the test region (T).
*NOTE: The intensity of the color in the test line region (T) will
vary depending on the concentration of HBsAg, HBsAb, HBeAg present
in the specimen. Therefore, any shade of color in the test region
(T) should be considered positive.
NEGATIVE: One colored line appears in the control region (C).No
apparent colored line appears in the test region (T).
INVALID: Control line fails to appear.Insufficient specimen volume
or incorrect procedural techniques are the most likely reasons for
control line failure. Review the procedure and repeat the test with
a new test cassette. If the problem persists, discontinue using the
test kit immediately and contact your local distributor.
HBeAb, HBcAb
NEGATIVE:* Two distinct colored lines appear. One colored line
should be in the control region (C) and another colored line should
be in the test region (T).
*NOTE: The intensity of the color in the test line region (T) may
vary. But it should be considered negative whenever there is even a
faint pink line.
Positive: One colored line appears in the control region (C).No
apparent colored line appears in the test region (T).
INVALID: Control line fails to appear.Insufficient specimen volume
or incorrect procedural techniques are the most likely reasons for
control line failure. Review the procedure and repeat the test with
a new test cassette. If the problem persists, discontinue using the
test kit immediately and contact your local distributor.
QUALITY CONTROL
A procedural control is included in the test. A red line appearing
in the control line region (C) is the internal procedural control.
It confirms sufficient specimen volume and correct procedural
technique.
Control standards are not supplied with this kit; however, it is
recommended that positive and negative controls be tested as good
laboratory practice to confirm the test procedure and to verify
proper test performance.
Order Information
| Cat. No. | Product | Specimen | Pack |
| IHB-355 | HBV Combo Rapid Test Cassette | S / P | 25 T |
| IHBSG-301 | HBsAg Rapid Test Dipstick | S / P | 50 T |
| IHBSG-302 | HBsAg Rapid Test Cassette | S / P | 40 T |
| IHBSG-401 | HBsAg Rapid Test Dipstick | WB/S/P | 50 T |
| IHBSG-402 | HBsAg Rapid Test Cassette | WB/S/P | 40 T |
| IHBSB-301 | HBsAb Rapid Test Dipstick | S / P | 50 T |
| IHBSB-302 | HBsAb Rapid Test Cassette | S / P | 40 T |
| IHBSB-401 | HBsAb Rapid Test Dipstick | WB/S/P | 50 T |
| IHBSB-402 | HBsAb Rapid Test Cassette | WB/S/P | 40 T |
| IHBCB-302 | HBcAb Rapid Test Cassette | S / P | 40 T |
| IHBEB-302 | HBeAb Rapid Test Cassette | S / P | 40 T |
| IHBEG-302 | HBeAg Rapid Test Cassette | S / P | 40 T |

15 Minutes HBV Combo Rapid Test Cassette HBsAg Rapid Test Dipstick
Inquiry Cart
0