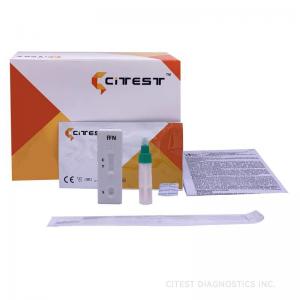
CE Fetal Fibronectin Rapid FFN Test 98.1% Sensitivity Home Rapid Test Kits
Add to Cart
Fetal Fibronectin (fFN) Rapid Test,,CE fetal fibronection At Home Rapid Diagnostic Test Kits
| Principle | Chromatographic Immunoassay |
| Format | Cassette |
| Specimen | Vaginal Secretion |
| Certificate | CE |
| Reading Time | 10 minutes |
| Pack | 25 T |
| Storage Temperature | 2-30°C |
| Shelf Life | 2 Years |
| Sensitivity | 98.10% |
| Specificity | 98.70% |
| Accuracy | 98.40% |
| Cut-Off | / |
Applications:
The Fetal fibronectin (fFN) Rapid Test Cassette (vaginal secretion) is a visually interpreted, qualitative immunochromatographic test device for the detection of fFN in cervicovaginal secretions to be used as an aid to assess the risk of pre-term delivery during pregnancy.
The test is intended for professional use to help diagnose the
rupture of fetal membranes (ROM) in pregnant women.
Description:
Fetal fibronectin (fFN), an isoform of fibronectin, is a complex adhesive glycoprotein with a molecular weight of approximately 500,000 daltons.
Fetal fibronectin is elevated in cervicovaginal secretions during
the first 24 weeks of pregnancy but is diminished between 24 and 34
weeks in normal pregnancies.Detection of fFN in cervicovaginal
secretions between 24 and 34 completed weeks gestation is reported
to be associated with preterm delivery in symptomatic and
asymptomatic pregnant women.
How to use?
Bring tests, specimens, buffer and/or controls to room temperature (15-30°C) before use.
1. Remove the test from its sealed pouch, and place it on a clean,
level surface. Label the test with patient or control
identification. To obtain a best result, the assay should be
performed within one hour.
2. Add 2 drops (80 ul) of the extracted buffer into the sample
well. As the test begins to work, you will see color move across
the membrane.
3. Wait for the colored band to appear. The result should be read at 10 minutes. Do not interpret the result after 20 minutes.
INTERPRETATION OF RESULTS
NOTE:
1. The intensity of the color in test region (T) may vary depending
on the concentration of aimed substances present in the specimen.
Therefore, any shade of color in the test region should be
considered positive. Besides, the substances level can not be
determined by this qualitative test.
2. Insufficient specimen volume, incorrect operation procedure, or
performing expired tests are the most likely reasons for control
band failure.
- For professional in vitro diagnostic use only. Do not use after the expiration date.
- Do not eat, drink or smoke in the area where the specimens and kits are handled.
- Handle all specimens as if they contain infectious agents. Observe established precautions against microbiological hazards throughout the procedure and follow the standard procedures for proper disposal of specimens.
- Wear protective clothing such as laboratory coats, disposable gloves and eye protection when specimens are assayed.
- The used test should be discarded according to local regulations.
- Humidity and temperature can adversely affect results.
- Do not use test if pouch is damaged