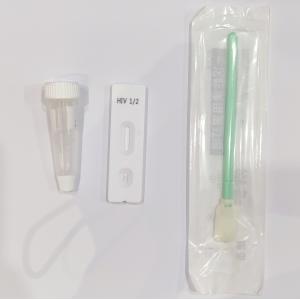
Product Details
HIV 1/2 AIDS Rapid Test Kit Near Gingival Oral Fluid For Human
Immunodeficiency Virus
| Storage | 2~30 º C |
| Specimen | Near Gingival Oral Fluid |
| Component | Rapid Test + Extraction Tube with Buffer + Swab |
| Principle | Colloidal gold rapid tests |
| Reading | Within 5mins |
| Package | 25tests/box; 1test/box |
| Trademark | Dewei |
| Origin | China |
INTENDED USE
The HIV 1/2 Rapid Test is a rapid visual immunoassay for the
qualitative, presumptive detection of antibodies to HIV-1/HIV-2 in
human near-gingival oral fluidspecimens. This kit is intended for
use as an aid in the diagnosis of HIV infection.
INTRODUCTION
HIV is the etiologic agent of Acquired Immune Deficiency Syndrome
(AIDS). The virion is surrounded by a lipid envelope that is
derived from host cell membrane. Several viral glycoproteins are on
the envelope. Each virus contains two copies of positive-sense
genomic RNAs. HIV-1 has been isolated from patients with AIDS and
AIDS-related complex, and from healthy people with high potential
risk for developing AIDS. HIV-2 has been isolated from West African
AIDS patients and from seropositive asymptomatic individuals.
PRINCIPLE
The HIV 1/2 Rapid Test detects antibodies to HIV-1/HIV-2 through
visual interpretation of color development on the internal strip.
Recombinant HIV antigens are immobilized on the test region of the
membrane. During testing, the specimen reacts with HIV antigen
conjugated to colored particles and precoated onto the sample pad
of the test. The mixture then migrates through the membrane by
capillary action and interacts with reagents on the membrane. If
there are sufficient HIV-1/HIV-2 antibodies in the specimen, a
colored band will form at the test region of the membrane. The
presence of this colored band indicates a positive result, while
its absence indicates a negative result. The appearance of a
colored band at the control region serves as a procedural control,
indicating that the proper volume of specimen has been added and
membrane wicking has occurred.
WARNINGS AND PRECAUTIONS
Immunoassay for in vitro diagnostic use only.
Do not use after expiration date.
The test should remain in the sealed pouch until use.
The used test should be discarded according to local regulations.
STORAGE AND STABILITY
The kit should be stored at 2-30°C until the expiry date printed on
the sealed pouch.
The test must remain in the sealed pouch until use.
Keep away from direct sunlight, moisture and heat.
Do not freeze.
Care should be taken to protect the components of the kit from
contamination. Do not use if there is evidence of microbial
contamination or precipitation. Biological contamination of
dispensing equipment, containers or reagents can lead to false
results.
OPERATION
Company Profile
DEWEI Medical Equipment Co., Ltd is a leading and professional
manufacturer of IVD products,it is a high-tech enterprise
specialized in developing, manufacturing and marketing of Clinical
Laboratory analyzer & reagents, such as Hematology Analyzer
& Reagents, Virus RNA Preservation & Extraction Kits, Urine Sediment Reagents and Washing Solutions.
DEWEI products are widely used in hospital clinics, disease control
centers, blood banks, veterinary clinics, scientific research
center and college laboratories. Qulity is primary concern, DEWEI team is strict on quality control
systems thus our products have CE, ISO, CFDA and other
international standards certificates.
The view of DEWEI is to be the most professional and respected
enterprise in IVD field, DEWEI will go on providing best service to
you by the outstanding R&D and cost-effective products.