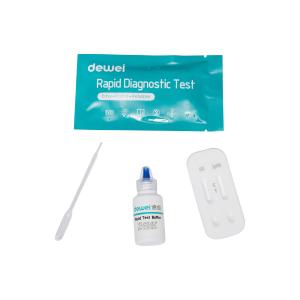
CE ISO 10 Minutes Syphilis Rapid Test Kit Cassette Type Whole Blood Serum Plasma Specimen
Add to Cart
CE ISO Syphilis Test Kit Rapid Syphilis Test Cassette Strip Whole Blood Serum Test
[INTENDED USE]
The Syphilis Rapid Test is a rapid visual immunoassay for the
qualitative, presumptive detection of IgM and IgG antibodies to
Treponema Pallidum (TP) in human whole blood, serum or plasma
specimens. This kit is intended for use as an aid in the diagnosis
of syphilis.
[CONTENTS]
• Rapid test cassette with desiccant
• Buffer
• Disposable pipettes
• Package insert
[OPERATION]
Bring tests, specimens, buffer and/or controls to room temperature
(15- 30°C) before use.
• Remove the test from its sealed pouch, and place it on a clean,
level surface. Label the device with patient or control
identification. For best results, the assay should be performed
within one hour.
• Using the provided disposable pipette, transfer 3 drops of
serum/plasma specimen (approximately 75 µL) to the specimen well
(S) of the device and start the timer.
OR
Transfer 2 drop of whole blood specimen (approximately 50 µL) to
the specimen well (S) of the device with the provided disposable
pipette, then add 1 drop of buffer and start the timer.
OR
Allow 2 hanging drops of fingerstick whole blood specimen to fall
into the center of the specimen well (S) on the device, then add 1
drop of buffer and start the timer. Avoid trapping air bubbles in
the specimen well (S), and do not add any solution to the result
area. As the test begins to work, you will see color move across
the membrane. • Wait for the colored band(s) to appear. The result
should be read at 10 minutes. Do not interpret the result after 20
minutes.
[INTERPRETATION OF RESULTS]
POSITIVE: The presence of two lines as control line (C) and test line (T)
within the result window indicates a positive result.The result
window indicates a positive result.
NEGATIVE: The presence of only control line (C) within the result window
indicates a negative result.
INVALID: If the control line (C) is not visible within the result window after performing the test, the result is considered invalid. Some causes of invalid results are because of not following the directions correctly or the test may have deteriorated beyond the expiration date. It is recommended that the specimen be re-tested using a new test.
[ABOUT US]
DEWEI Medical Equipment Co., Ltd is a leading and professional
manufacturer of IVD products,it is a high-tech enterprise
specialized in developing, manufacturing and marketing of Clinical
Laboratory analyzer & reagents, such as Hematology Analyzer
& Reagents, Virus RNA Preservation & Extraction Kits, Urine
Sediment Reagents and Washing Solutions.
DEWEI products are widely used in hospital clinics, disease control
centers, blood banks, veterinary clinics, scientific research
center and college laboratories.
Qulity is primary concern, DEWEI team is strict on quality control
systems thus our products have CE, ISO, CFDA and other
international standards certificates.
The view of DEWEI is to be the most professional and respected
enterprise in IVD field, DEWEI will go on providing best service to
you by the outstanding R&D and cost-effective products.